 Both these molecules contain C, H and O atoms. The hybridization of carbon in the carbonyl group is sp 2.Įthers and ketones are organic molecules. The hybridization of carbon in C-O-C bond is sp 3. Less acidic than a ketone but is highly acidic than hydrocarbons. Ketone is an organic compound that contains an oxygen atom bonded to a carbon atom via a double bond. This carbon atom is sp 2 hybridized.Įther is an organic compound that contains two alkyl groups bonded to the same oxygen atom. 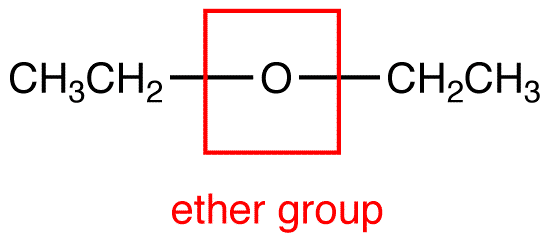
The central carbon atom along with the double bonded oxygen atom forms the carbonyl group. The R groups indicate the alkyl or aryl groups. Here, the bonding between the oxygen atom and the carbon atom is a double bond. What is Ketone?Ī ketone is an organic molecule having the chemical formula R-C-(=O)R. And also ethers are slightly polar because of the bond angle of the C-O-C bond. However, they can form hydrogen bonds with water molecules because there are lone electron pairs on the oxygen atom. This results in lower boiling points because there are no strong interaction forces between its molecules. However, it is less acidic than that of carbonyl compounds such as ketones.Įthers cannot form hydrogen bonds with each other. That means, the hydrogen atom bonded to the carbon atom and is adjacent to the C-O-C bond releases easily in the from of a proton. Since the oxygen atom is more electronegative than the carbon atom, the alpha hydrogen of an ether is highly acidic in comparison to a hydrocarbon. 
The hybridization of each carbon of this functional group is sp 3. Therefore, it acts as the functional group. The C-O-C chemical bond that has a 110° bond angle decides the characteristics of an ether. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
